WITH BRAM-COR TECHNOLOGIES FROM DESIGN TO VALIDATION
Thanks to a fruitful experience in many branches of making pharmaceuticals, BRAM-COR has matured a coherent path, able to face meaningful business opportunities for customers who wish to diversify or set up production lines of parenteral solutions, both for bags and for bottles, within turnkey or segmented projects.

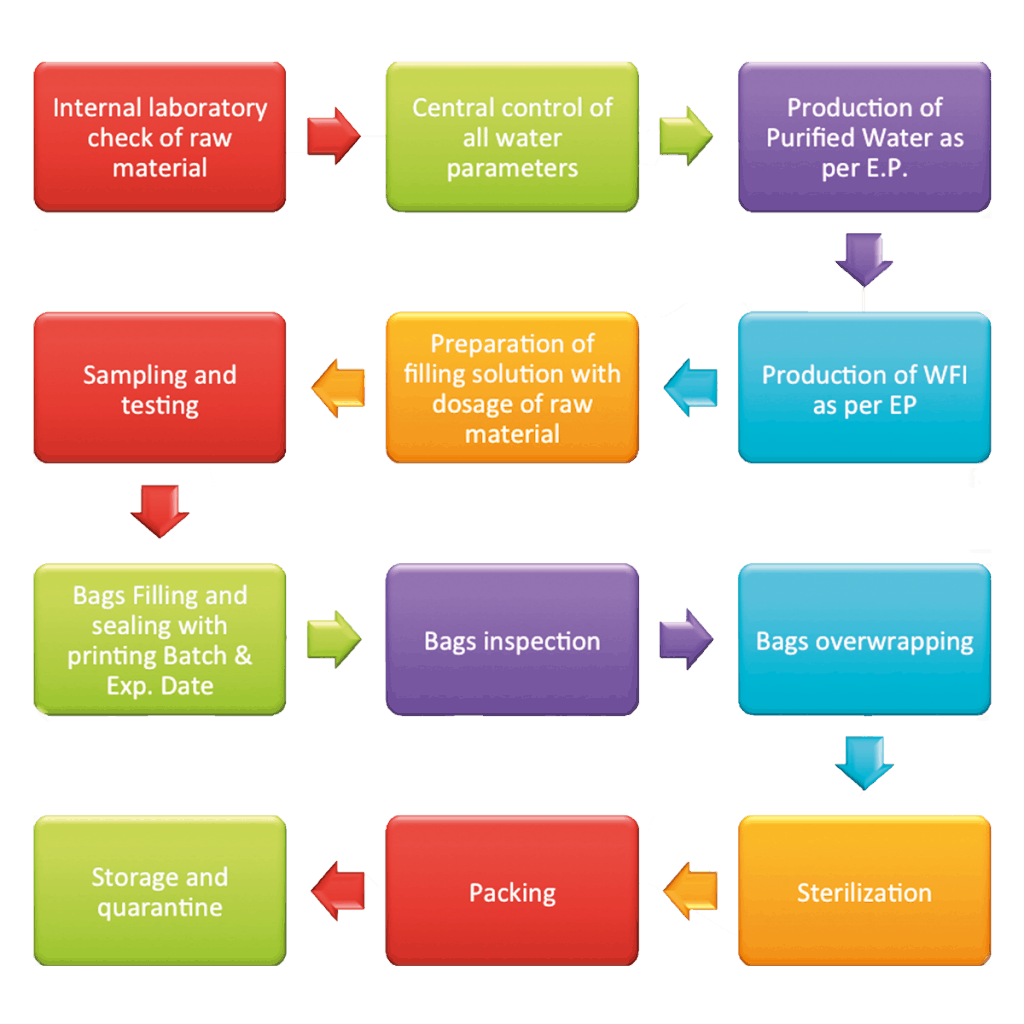
Our specific knowledge begins from water and from its transformation into a basic element for the pharmaceutical process, with a trouble-free production stages in storage and distribution, processing and filling. This holistic vision is a key strategy to obtain flexibility, efficiency and convenience in small or large volume parenterals, and in a wide range of products:
– STANDARD IVF & ELECTROLITES
– DIALYSIS SOLUTIONS
– TPN (Total Parenteral Nutrition)
– SPECIAL SOLUTIONS
In our turnkey offers we usually exlude those items that can be procured locally at reasonable prices by the customer itself (like land, buildings, black utilities, …).
Of course we therefore provide, on demand :
– Advanced Know How Transfer for additional IV solutions, TPN, etc.
– Post-Start up assistance
– Raw Materials and Consumables
– Black Utilities
A TURNKEY ROADMAP
The standards of good manufacturing practice (cGMP) require special attention to risk assessment and verification procedures: “… it is requirement of good manufacturing identify the activities of validation necessary to demonstrate control critical aspects of particular operations. The significant changes made to installations, equipment and processes, which may affect product quality, should be validated. A procedure for risk assessment should be used to determine the scope and extent of validation.”
The Validation Master Plan serves to make sure that all equipment, procedures, that may affect the quality or integrity or effectiveness of the product, are validated; it contains the general principles which comply during the validation task, and plans activities to be carried out for this purpose.
√ Basic Engineering
√ Detailed Engineering
√ Design Qualification
√ Inlet Water Pretreatment Plant
√ Pharmaceutical Water Systems (Softened, Purified and Distilled Water)
√ Pharmaceutical Processing and Solution Preparation Systems
√ Pharmaceutical Forming, Filling, Inspecting, Packaging lines
√ Clean Rooms
√ Epoxy coating of the floors
√ HVAC and air treatment plant
√ Autoclave
√ Pure Steam Generator and PS circuit
√ Laboratories of Analysis (Microbiological / Chemical)
√ Site Master Plan
√ Validation Master Plan
√ Installation
√ Training
√ Start up
√ Technical Files & Documentation
√ IQ/OQ
√ PQ Protocols
√ Validation at Site
√ Standard Operating Procedures
√ Initial Know How Transfer
√ GMP preAudit
√ Spareparts for n years